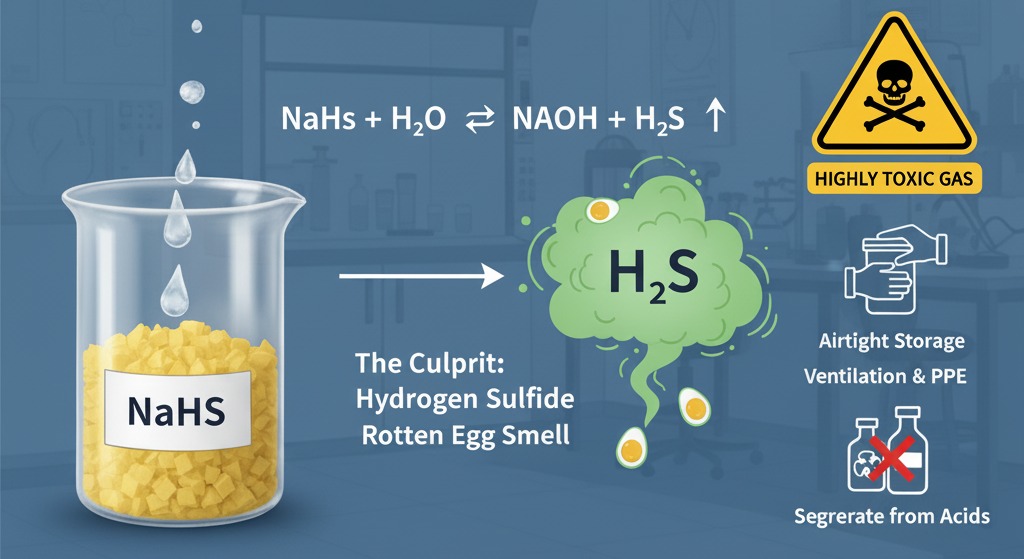
Sodium Hydrosulfide (NaHS) is widely used in leather processing, mineral flotation, dye synthesis, pharmaceuticals, and wastewater treatment. However, professionals handling NaHS solutions often notice a sharp, unpleasant odor—commonly described as a “rotten egg smell.”
What causes this distinctive scent? The answer lies in chemistry—and safety.
This article explains the science behind the odor, how hydrogen sulfide forms, and why proper handling of sodium hydrosulfide is essential in industrial environments.
The Real Cause: Hydrogen Sulfide (H₂S)
The characteristic rotten egg smell associated with NaHS comes from hydrogen sulfide (H₂S).
Hydrogen sulfide is:
-
A colorless gas
-
Highly toxic
-
Flammable
-
Recognizable by its strong sulfur odor
It naturally occurs in volcanic emissions and during the anaerobic decomposition of organic matter. In industrial settings, H₂S is classified as a hazardous gas due to its toxicity and rapid health effects.
According to the United States Environmental Protection Agency, hydrogen sulfide is considered a hazardous air pollutant requiring strict exposure control.
How Sodium Hydrosulfide Releases Hydrogen Sulfide
Pure, dry sodium hydrosulfide flakes are theoretically odorless. The smell develops when NaHS reacts with moisture, air, or acids.
Below are the primary chemical pathways responsible for H₂S release.
1. Hydrolysis in Water (Primary Source)
When NaHS dissolves in water—or absorbs moisture from humid air—it undergoes hydrolysis:
NaHS + H₂O ⇌ NaOH + H₂S↑
This equilibrium reaction continuously releases small amounts of hydrogen sulfide gas, which creates the detectable odor.
Even trace levels of H₂S are detectable due to the human nose’s high sensitivity to sulfur compounds.
2. Reaction with Carbon Dioxide (CO₂) in Air
Carbon dioxide in the atmosphere forms weak carbonic acid when dissolved in moisture. This slight acidity can trigger H₂S release:
NaHS + CO₂ + H₂O → NaHCO₃ + H₂S↑
Although slower than acid reactions, this process contributes to the persistent low-level odor in storage areas.
3. Reaction with Strong Acids (High-Risk Scenario)
When sodium hydrosulfide contacts stronger acids, the reaction becomes rapid and potentially dangerous:
NaHS + Acid → H₂S↑ (rapid release)
This can generate large volumes of hydrogen sulfide gas in a short time, posing severe inhalation hazards.
⚠ Acid contact with NaHS can cause sudden, high-concentration H₂S exposure.
Why the Smell Is a Critical Warning Signal
The rotten egg smell is more than just unpleasant—it serves as an early warning sign.
However, hydrogen sulfide has a dangerous property: at high concentrations, it can cause olfactory fatigue (temporary loss of smell). This means:
-
Workers may stop detecting the odor
-
Dangerous gas levels may go unnoticed
-
Risk of acute poisoning increases
Because of this, smell alone must never be relied upon as a safety indicator.
Health Risks of Hydrogen Sulfide Exposure
Hydrogen sulfide exposure may cause:
-
Eye and respiratory irritation
-
Headaches and dizziness
-
Nausea
-
Shortness of breath
-
Loss of consciousness at high concentrations
Industrial facilities must implement strict monitoring and ventilation systems to manage exposure.
Safety Protocols for Handling Sodium Hydrosulfide
To minimize H₂S release and exposure risks, follow these essential safety measures:
1. Airtight Storage
-
Store NaHS in tightly sealed containers
-
Prevent moisture ingress
-
Use corrosion-resistant packaging

2. Proper Ventilation
-
Operate in well-ventilated areas
-
Use local exhaust ventilation (LEV)
-
Install fixed H₂S gas detectors
-
Conduct regular air monitoring
3. Personal Protective Equipment (PPE)
-
Chemical-resistant gloves
-
Safety goggles or face shield
-
Protective clothing
-
Respirators in confined spaces
4. Chemical Segregation
-
Keep NaHS strictly separated from acids
-
Store away from incompatible materials
-
Label storage areas clearly
5. Emergency Planning
-
Establish H₂S leak response protocols
-
Conduct emergency drills
-
Maintain evacuation procedures
-
Install eyewash stations and safety showers
Preparedness significantly reduces operational risk.
COTEC’s Commitment to NaHS Safety
As a professional sodium hydrosulfide supplier, COTEC prioritizes product stability and safe handling.
With over 30 years of experience in hazardous chemical management, the company implements:
-
Strict production controls
-
Moisture-controlled packaging
-
Careful transportation standards
-
Batch quality inspections
These measures help minimize premature hydrogen sulfide generation and ensure consistent product purity.
For detailed Safety Data Sheets (SDS) or professional guidance on sodium hydrosulfide storage and usage, consulting experienced suppliers is strongly recommended.
Conclusion
The “rotten egg smell” associated with sodium hydrosulfide is caused by the release of hydrogen sulfide gas. This occurs when NaHS reacts with moisture, carbon dioxide, or acids.
Understanding this chemical mechanism is essential for:
-
Industrial safety
-
Proper storage
-
Regulatory compliance
-
Environmental protection
By implementing airtight storage, ventilation systems, gas monitoring, and strict segregation from acids, industries can safely manage sodium hydrosulfide while minimizing hydrogen sulfide risks.
The odor is not just unpleasant—it is a chemical warning that demands respect and proper safety protocols.